✅ "Pharmacy-Trusted Since 2013."
Does Honey Ever Expire?
The Science of Why Pure Honey Doesn't Spoil!
Pure honey is a "bioactive" powerhouse with a unique chemical composition that prevents the growth of bacteria and fungi. Its eternal shelf life is driven by three primary factors: low moisture, high acidity (pH 3.4 to 6.1), and the production of hydrogen peroxide.
How Bees Create a Natural Preservative
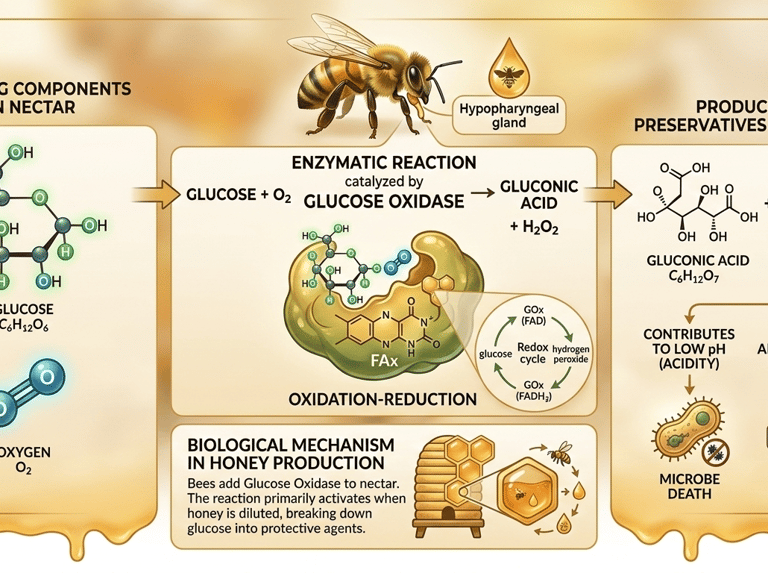
Hydrogen peroxide does not exist in flower nectar; it is created during the honey-making process. Worker bees secrete an enzyme called glucose oxidase from their hypopharyngeal glands and add it to the collected nectar.
The Glucose Oxidase Reaction
Through a specific oxidation reaction, glucose oxidase catalyses the conversion of glucose into gluconic acid and hydrogen peroxide. This reaction activates when honey is slightly diluted, lowering its osmotic pressure.
The biochemical process can be summarised by this formula:
Glucose + O₂ → Gluconic Acid + H₂O.
Gluconic Acid: Increases the honey's acidity, creating a hostile environment for microbes.
Hydrogen Peroxide: Provides the potent antimicrobial and therapeutic properties that define medicinal honey.
This elegant biochemistry transforms simple nectar into a complex, shelf-stable food that has been valued for its healing properties across cultures for millennia.
In Summary
The unique chemical composition and natural properties of pure honey prevent the growth of microorganisms, thereby preventing spoilage. The primary factors preventing bacteria and fungi from thriving in pure honey include low moisture content, its high acidity (pH 3.4 to 6.1), and the antimicrobial chemical component hydrogen peroxide.
Hydrogen peroxide does not naturally occur in nectar. The enzyme responsible for hydrogen peroxide production in honey is glucose oxidase. Glucose oxidase is an enzyme secreted by the hypopharyngeal glands of worker bees. Bees add this enzyme to the collected nectar during the honey-making process. Glucose oxidase is also involved in several other biochemical changes that occur in nectar as the honey forms.
The conversion of glucose to hydrogen peroxide is a specific oxidation reaction catalysed by the glucose oxidase enzyme. In this reaction, the enzyme catalyses the oxidation of glucose (C₆H₁₂O₆) to gluconic acid (C₆H₁₂O₇) while simultaneously reducing the molecular oxygen (O₂) to hydrogen peroxide (H₂O₂). This reaction can be summed up as: glucose + O₂ → gluconic acid + H₂O₂. The reaction proceeds when honey is diluted (which reduces its osmotic pressure and increases the water activity). The resulting gluconic acid is one of the factors contributing to the acidity of honey, while hydrogen peroxide contributes to the antimicrobial properties of honey.
The formation of hydrogen peroxide in honey from glucose via the enzyme glucose oxidase is an elegant example of nature's biochemistry. This process not only preserves honey but also contributes to its therapeutic properties. This understanding of the processes which occur during the formation of honey helps us to better understand and appreciate honey as not just a sweetener, but as a complex bioactive food that has been enjoyed by humans across cultures and throughout history.


Does Honey Ever Expire?
Pure honey is a "bioactive" powerhouse with a unique chemical composition that inhibits the growth of bacteria and fungi. Its remarkable shelf life is driven by three primary factors: low moisture content, high acidity (with a pH range of 3.4 to 6.1), and the production of hydrogen peroxide. Together, these elements ensure that pure honey remains safe and stable over time. An amazing biochemistry transforming simple nectar into a complex, shelf-stable food that has been valued for its healing properties across cultures for millennia.
The Science: Why Pure Honey Doesn't Spoil!
How Bees Create a Natural Preservative
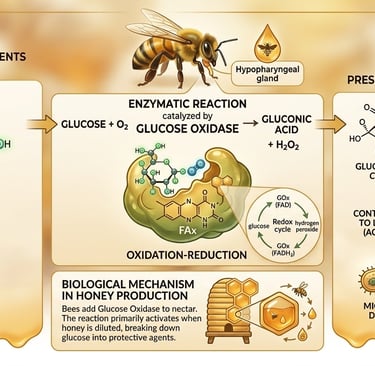
Hydrogen peroxide does not exist in flower nectar; it is created during the honey-making process. Worker bees secrete an enzyme called glucose oxidase from their hypopharyngeal glands and add it to the collected nectar. Glucose oxidase is also involved in other biochemical changes that occur in nectar as the honey forms.
The Glucose Oxidase Reaction
Through a specific oxidation reaction, glucose oxidase catalyses the conversion of glucose into gluconic acid and hydrogen peroxide. This reaction activates when honey is slightly diluted, lowering its osmotic pressure.
The biochemical process can be summarised by the following equation: Glucose + O₂ → Gluconic Acid + H₂O.
Gluconic Acid: Increases the honey's acidity, creating a hostile environment for microbes.
Hydrogen Peroxide: Provides the potent antimicrobial and therapeutic properties that define medicinal honey.
Biochemical mechanism of glucose oxidase in honey production: A step-by-step flowchart of the enzymatic oxidation-reduction cycle from bee hypopharyngeal glands.
The Science: Why Pure Honey Doesn't Spoil!
Pure honey is a "bioactive" powerhouse with a unique chemical composition that inhibits the growth of bacteria and fungi. Its remarkable shelf life is driven by three primary factors: low moisture content, high acidity (with a pH range of 3.4 to 6.1), and the production of hydrogen peroxide. Together, these elements ensure that pure honey remains safe and stable over time.
This sophisticated biochemical conversion transforms raw nectar into a bioactive superfood, prized across global cultures for millennia due to its natural antimicrobial properties and unparalleled shelf stability.
The Glucose Oxidase Reaction
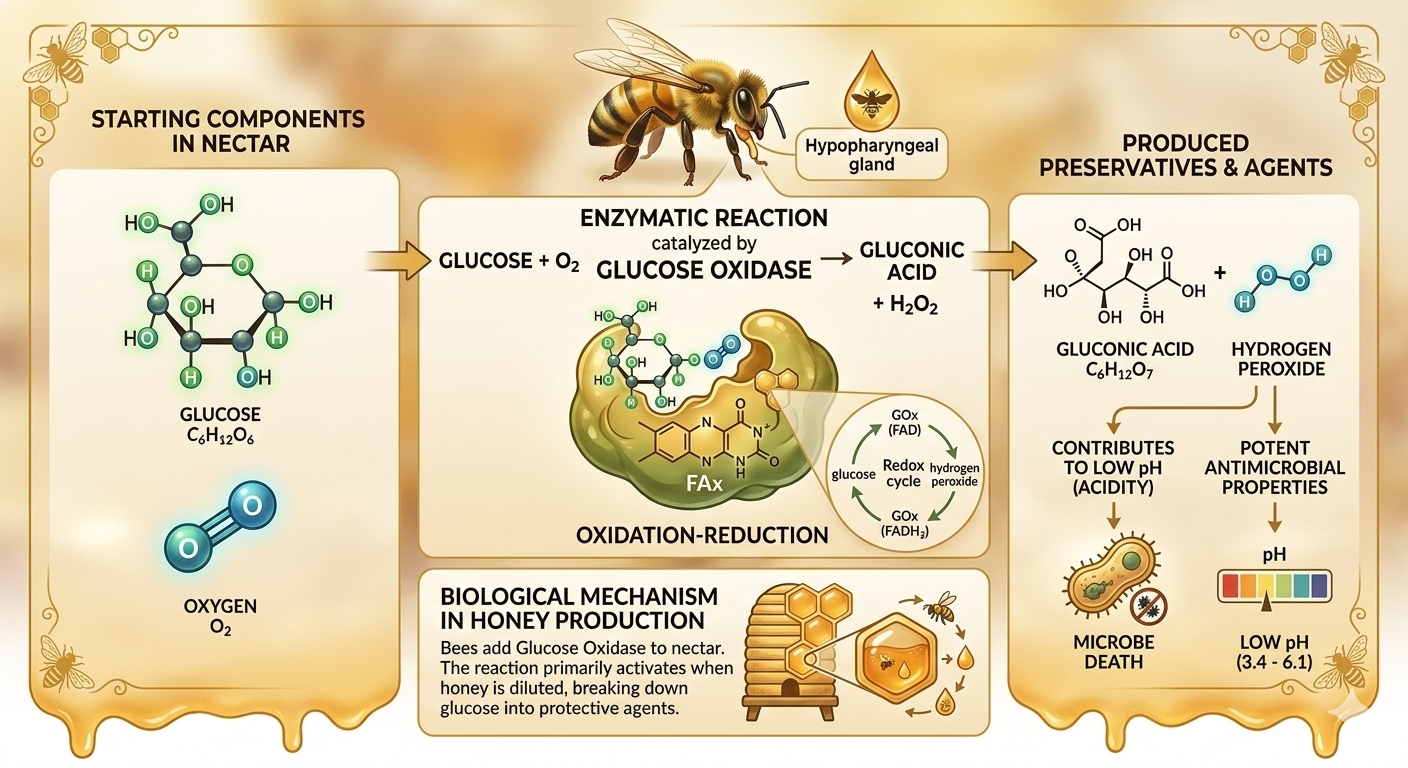
Through a sophisticated oxidation-reduction process, the enzyme glucose oxidase catalyses the transformation of glucose into gluconic acid and hydrogen peroxide. This natural preservation mechanism activates as honey is slightly diluted, which lowers the osmotic pressure and triggers the following biochemical reaction:
Glucose + O₂ → Gluconic Acid + H₂O.
Gluconic Acid: Increases the honey's acidity, creating a hostile environment for microbes.
Hydrogen Peroxide: Provides the potent antimicrobial and therapeutic properties that define medicinal honey.
The Enzymatic Genesis of Honey Preservation
While flower nectar is a nutrient-rich energy source, it is also highly perishable and lacks the antimicrobial potency of finished honey. This vital transition occurs through a sophisticated biological intervention. As foraging worker bees collect nectar, they introduce glucose oxidase—a specialised enzyme synthesised in their hypopharyngeal glands.
This enzyme acts as a biological "time-release" preservative. Once added to the nectar, glucose oxidase initiates a series of complex biochemical changes during the ripening process within the hive. In the presence of oxygen, the enzyme begins the slow oxidation of glucose. This doesn't just create a single chemical; it fundamentally alters the nectar's molecular profile, lowering its pH and preparing it for long-term storage.
Pure honey is a "bioactive" powerhouse with a unique chemical composition that inhibits the growth of bacteria and fungi. Its remarkable shelf life is driven by three primary factors: low moisture content, high acidity (with a pH range of 3.4 to 6.1), and the production of hydrogen peroxide. Together, these elements ensure that pure honey remains safe and stable over time.
This sophisticated biochemical conversion transforms raw nectar into a bioactive superfood, prized across global cultures for millennia due to its natural antimicrobial properties and unparalleled shelf stability.
The Glucose Oxidase Reaction
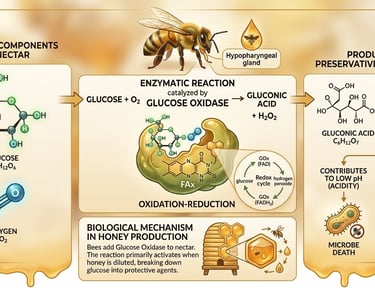
Through a sophisticated oxidation-reduction process, the enzyme glucose oxidase catalyses the transformation of glucose into gluconic acid and hydrogen peroxide. This natural preservation mechanism activates as honey is slightly diluted, which lowers the osmotic pressure and triggers the following biochemical reaction:
Glucose + O₂ → Gluconic Acid + H₂O.
Gluconic Acid: Increases the honey's acidity, creating a hostile environment for microbes.
Hydrogen Peroxide: Provides the potent antimicrobial and therapeutic properties that define medicinal honey.
How bees make hydrogen peroxide and the Enzymatic Genesis of Honey Preservation
While flower nectar is a nutrient-rich energy source, it is also highly perishable and lacks the antimicrobial potency of finished honey. Hydrogen peroxide does not exist in flower nectar; it is created during the honey-making process. Foraging worker bees secrete an enzyme called glucose oxidase from their hypopharyngeal glands and add it to the collected nectar. Glucose oxidase also plays a role in other biochemical changes that occur in nectar during the formation of honey.
Biochemical mechanism of glucose oxidase in honey production: A step-by-step flowchart of the enzymatic oxidation-reduction cycle from bee hypopharyngeal glands.


Biochemical mechanism of glucose oxidase in honey production: A step-by-step flowchart of the enzymatic oxidation-reduction cycle from bee hypopharyngeal glands.
The Science: Why Pure Honey Doesn't Spoil!
Pure honey is a "bioactive" powerhouse with a unique chemical composition that inhibits the growth of bacteria and fungi. Its remarkable shelf life is driven by three primary factors: low moisture content, high acidity (with a pH range of 3.4 to 6.1), and the production of hydrogen peroxide. Together, these elements ensure that pure honey remains safe and stable over time.
This sophisticated biochemical conversion transforms raw nectar into a bioactive superfood, prized across global cultures for millennia due to its natural antimicrobial properties and unparalleled shelf stability.


The Glucose Oxidase Reaction
Through a sophisticated oxidation-reduction process, the enzyme glucose oxidase catalyses the transformation of glucose into gluconic acid and hydrogen peroxide. This natural preservation mechanism activates as honey is slightly diluted, which lowers the osmotic pressure and triggers the following biochemical reaction:
Glucose + O₂ → Gluconic Acid + H₂O.
Gluconic Acid: Increases the honey's acidity, creating a hostile environment for microbes.
Hydrogen Peroxide: Provides the potent antimicrobial and therapeutic properties that define medicinal honey.
A short story about us
Pure honey is a "bioactive" powerhouse with a unique chemical composition that inhibits the growth of bacteria and fungi. Its remarkable shelf life is driven by three primary factors: low moisture content, high acidity (with a pH range of 3.4 to 6.1), and the production of hydrogen peroxide. Together, these elements ensure that pure honey remains safe and stable over time.
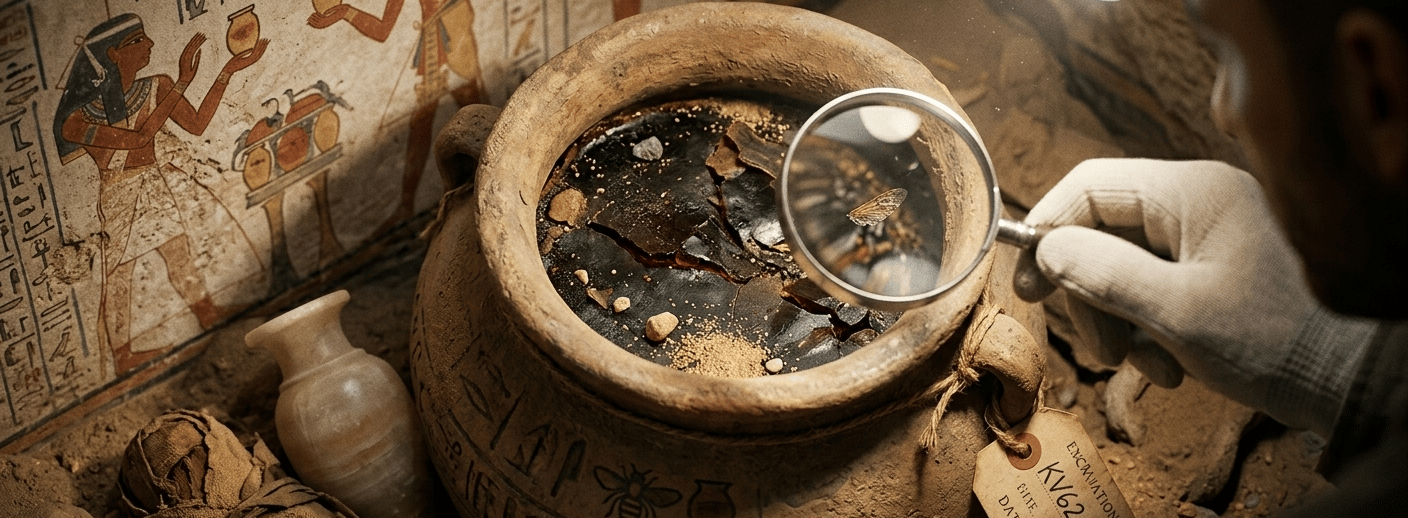

The Science: Why Pure Honey Doesn't Spoil!
How Bees Convert Glucose into Hydrogen Peroxide
The conversion of glucose to hydrogen peroxide is a specific oxidation reaction catalysed by the glucose oxidase enzyme. In this reaction, the enzyme catalyses the oxidation of glucose (C₆H₁₂O₆) to gluconic acid (C₆H₁₂O₇) while simultaneously reducing the molecular oxygen (O₂) to hydrogen peroxide (H₂O₂). This reaction can be summed up as: glucose + O₂ → gluconic acid + H₂O₂. The reaction proceeds when honey is diluted (which reduces its osmotic pressure and increases the water activity). The resulting gluconic acid is one of the factors contributing to the acidity of honey, while hydrogen peroxide contributes to the antimicrobial properties of honey.
The formation of hydrogen peroxide in honey from glucose via the enzyme glucose oxidase is an elegant example of nature's biochemistry. This process not only preserves honey but also contributes to its therapeutic properties. This understanding of the processes which occur during the formation of honey helps us to better understand and appreciate honey as not just a sweetener, but as a complex bioactive food that has been enjoyed by humans across cultures and throughout history.
Did You Know? The "Wound Care" Secret
This biochemical reaction is exactly why honey has been used as a medical dressing for thousands of years. When raw honey comes into contact with the fluid from a wound (which is mostly water), the glucose oxidase "wakes up" and begins producing a slow, steady release of hydrogen peroxide.
Unlike the bottled hydrogen peroxide from a pharmacy—which is high-strength and can actually damage healthy tissue—the peroxide produced by honey is delivered in "micro-doses." This is strong enough to kill bacteria but gentle enough to promote the growth of new skin cells.
A short story about us
Pure honey is a "bioactive" powerhouse with a unique chemical composition that inhibits the growth of bacteria and fungi. Its remarkable shelf life is driven by three primary factors: low moisture content, high acidity (with a pH range of 3.4 to 6.1), and the production of hydrogen peroxide. Together, these elements ensure that pure honey remains safe and stable over time.
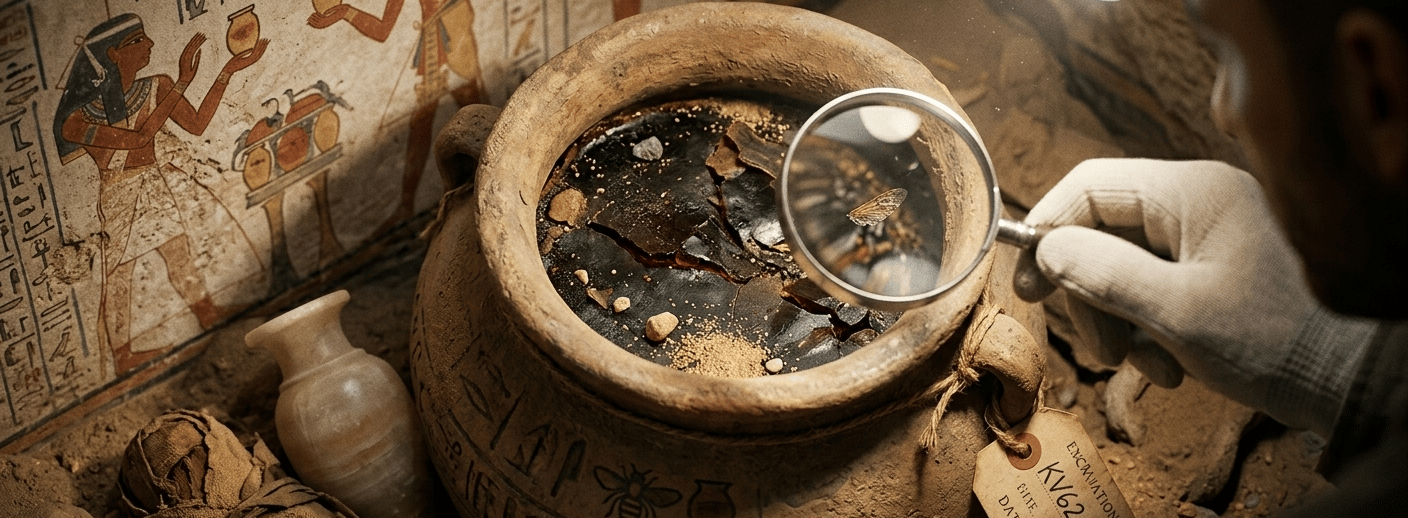

The Science: Why Pure Honey Doesn't Spoil!
The Bioactive Conversion of Glucose to Hydrogen Peroxide
The conversion of glucose to hydrogen peroxide is a specific oxidation reaction catalysed by the glucose oxidase enzyme. In this reaction, the enzyme catalyses the oxidation of glucose (C₆H₁₂O₆) to gluconic acid (C₆H₁₂O₇) while simultaneously reducing the molecular oxygen (O₂) to hydrogen peroxide (H₂O₂). This reaction can be summed up as: glucose + O₂ → gluconic acid + H₂O₂. The reaction proceeds when honey is diluted (which reduces its osmotic pressure and increases the water activity). The resulting gluconic acid is one of the factors contributing to the acidity of honey, while hydrogen peroxide contributes to the antimicrobial properties of honey.
The formation of hydrogen peroxide in honey from glucose via the enzyme glucose oxidase is an elegant example of nature's biochemistry. This process not only preserves honey but also contributes to its therapeutic properties. This understanding of the processes which occur during the formation of honey helps us to better understand and appreciate honey as not just a sweetener, but as a complex bioactive food that has been enjoyed by humans across cultures and throughout history.
Did You Know? The "Wound Care" Secret
This biochemical reaction is exactly why honey has been used as a medical dressing for thousands of years. When raw honey comes into contact with the fluid from a wound (which is mostly water), the glucose oxidase "wakes up" and begins producing a slow, steady release of hydrogen peroxide.
Unlike the bottled hydrogen peroxide from a pharmacy—which is high-strength and can actually damage healthy tissue—the peroxide produced by honey is delivered in "micro-doses." This is strong enough to kill bacteria but gentle enough to promote the growth of new skin cells.
Can Real Honey Go Bad? The Truth About Storage and Shelf Life
Does Honey Expire? The Science of Moisture and Fermentation
Why Honey is "Hygroscopic"
Honey is hygroscopic, meaning it acts like a sponge for the environment. It actively attracts and retains moisture from the air and can even absorb surrounding odours. While this makes honey unique, it is also the primary reason real honey can spoil if not stored in an airtight container.
The 20% Rule: The Critical Limit for Honey Safety
To maintain its "eternal" shelf life, honey must stay below a 20% moisture threshold. This specific concentration is the "magic number" for two reasons:
Osmotic Pressure: At moisture levels below 20%, the osmotic pressure is too high for yeast cells to survive. They essentially dehydrate and remain dormant.
Preventing Fermentation: Once moisture exceeds 20%, the osmotic pressure drops. This allows yeast cells to become active, triggering fermentation and altering the taste, smell, and safety of the honey.
Pro Tip: Always keep your honey lid tightly sealed. Because of its hygroscopic nature, even a humid kitchen can push your honey past the 20% limit over time.


How to Store Honey: The Ultimate 2026 Guide for Eternal Shelf Life
If your honey has turned solid and white, it is not spoiled. Crystallisation is a natural process. To reliquefy it, place the tightly sealed jar in a bowl of warm water (not boiling, max 40°C / 104°F) and let it sit until the crystals dissolve.
The Critical Temperature Thresholds
33°C – 36°C (91.4°F – 96.8°F): This is approximately the average temperature of a natural beehive. Once honey exceeds 40°C / 104°F, it begins to lose its delicate enzymes and nutritional complexity.
Above 60°C (140°F): This is the "danger zone." Exposure to these temperatures rapidly destroys antioxidants, nutrients, antimicrobial properties, and enzymes like glucose oxidase.
Because honey is hygroscopic (it absorbs moisture from the air), proper storage is essential to keep its moisture content below the critical 20% threshold. Follow this checklist to ensure your honey never spoils.
[ ] Seal it Tight (Always): This is the most critical step. Use an airtight container (glass or high-quality plastic). A loose lid allows honey to absorb humidity from the air, raising its moisture level and risking fermentation.
[ ] Store in a Cool, Dry Place: The ideal location is a pantry or cupboard away from direct heat and sunlight. The ideal temperature is between 21°C–27°C (70°F–80°F). Avoid storing honey near the stove or dishwasher.
[ ] Refrigeration: While you can refrigerate honey, it will accelerate crystallisation (turning it hard and grainy). While crystallised honey isn't spoiled, it is harder to use. Keep it on the shelf instead.
[ ] Use Clean, Dry Utensils: Never put a wet spoon or a utensil with food residue (like breadcrumbs) back into the honey jar. This introduces external moisture and contaminants that can trigger mould or bacterial growth.
The Science of Honey Texture: How Refrigeration Impacts Different Varietals
The crystallisation of honey depends on its sugar profile. Varietals with a high glucose-to-fructose ratio crystallise quickly, whereas high-fructose honeys usually remain clear but can harden into a dense, toffee-like consistency when stored in the fridge.
What about Crystallisation?
Honey Storage Checklist: 4 Steps to Prevent Spoilage


From Pharaohs to Modern Labs: The Timeless Properties of Raw Honey
You didn’t come this far to stop


3,000 Years of Healing: The History and Longevity of Pure Honey
Archaeologists have uncovered jars of honey in ancient Egyptian tombs that remain perfectly preserved and edible after more than 3,000 years. This incredible longevity is due to honey's unique chemical makeup—a combination of low moisture content, high acidity (pH between 3 and 4.5), and the presence of natural hydrogen peroxide that creates an environment where bacteria simply cannot survive.
Beyond its shelf life, honey has been a cornerstone of global medicine for millennia; the Sumerians recorded its use as a drug as early as 2000 BC, while Aristotle and Hippocrates later prescribed specific varieties for wound healing and respiratory health. This "ancient power" is now validated by modern science, confirming that the same enzymes found in the Pharaohs' tombs are what provide honey with its potent antimicrobial and antioxidant properties today.
The Mystery of King Tutankhamun’s Honey
The 1922 excavation of King Tutankhamun’s tomb provided the world with a fascinating look at honey’s eternal shelf life. While popular myths suggest the honey was still liquid and "edible," archaeologists actually discovered dehydrated honey residue and beeswax. These findings prove that honey’s unique chemical properties—low moisture and high acidity—allow it to survive for millennia, even if its texture changes over time.
Ancient Egypt: The World's First Commercial Beekeepers
In Ancient Egypt, beekeeping was more than a hobby; it was a state-controlled industry. Egyptians were among the first to practice migratory beekeeping, moving clay hives on rafts along the Nile to follow seasonal flower blooms.
Sustenance & Ritual: Honey served as a primary culinary sweetener and a powerful medicinal salve.
Divine Offerings: Often referred to as "Liquid Gold," honey was an essential offering for the afterlife, symbolised by the bee icon representing Lower Egypt.
Tel Rehov: The 3,000-Year-Old Industrial Apiary
Further evidence of ancient honey production was discovered at Tel Rehov, Israel. This 3,000-year-old site revealed the oldest known industrial apiary in the world.
Advanced Systems: The discovery of over 30 intact clay hives suggests a massive operation capable of producing half a ton of honey annually.
Global Trade: DNA analysis of bee remains at the site indicates that these ancient beekeepers may have even imported bees from Turkey to ensure higher yields and calmer temperaments.From the pharaohs of Egypt to the organized apiaries of Israel, honey remains a tangible link to our ancestors. It is a rare food that connects modern culinary traditions directly to the agricultural mastery of historical societies.
Melli Magic Honey®
+61-411234759
Melli Magic Honey® © 2026. All rights reserved.
ABN: 65366959676
149 The Grand Parade, Monterey, NSW, 2217
